For decades, scientists and clinicians have been puzzled by an autoimmune condition, antiphospholipid syndrome (APS), which causes devastating health problems including uncontrolled blood clots and repeated pregnancy loss. Recent studies have shown that naturally produced antibodies that bind to phosphatidylethanolamine (PE) are positively correlated to antiphospholipid syndrome.
Phosphatidylethanolamine (PE), a lipid that makes up 20% of the inner cell membrane, has roles in membrane trafficking and reorganization. Paradoxically, the antibodies that bind to PE (anti-PE antibodies) in APS patients are produced outside of cells. This raises the question of how do the anti-PE antibodies bind to PE without having access to their targets?
To unravel this mystery, Chemistry of Life Processes Institute faculty member and Northwestern Medicine investigator Ming Zhao (cardiology) synthesized special molecular probes to investigate the dynamic distribution of PE in living cells under the microscope. The probes were designed to bind to PE and visualize its location by way of fluorescence. Zhao and research associate Songwang Hou worked with colleagues Heike Folsch (cell and molecular biology), Joan Cook Mills (allergy-immunology/medicine), and Rosalind Ramsey-Goldman (rheumatology/medicine) to study the mechanism of anti-PE antibody binding.
The study was published in a PNAS paper on December 26, 2017. Zhao was the senior author on the publication.
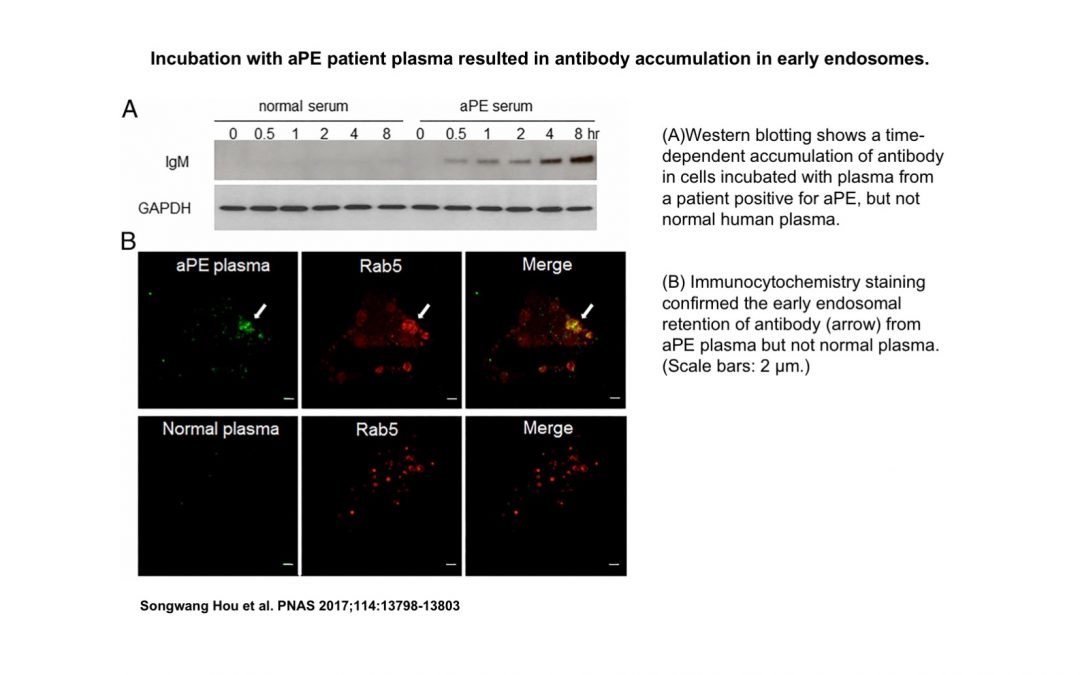
The authors verified that the cells’ surface membranes did not contain PE, consistent with the lack of binding targets for the anti-PE antibodies. But, to their surprise, they discovered PE is accessible to the anti-PE antibodies through a subcellular structure called the endosome.
Endosomes are membrane-bound vesicles that form a pocket-like structure to the cells’ exterior environment, where they absorb outside materials and allow the cell to interact with its environment. The membrane encasing the endosome contains PE.
When outside material enters these pockets, the endosome wraps around it, transforming into a vesicle, a sac-like structure, to carry the material into the cell. It is at this transient moment that PE becomes accessible to outside materials, including the anti-PE antibodies.
Zhao’s data demonstrated that binding of anti-PE antibodies to PE in the vesicles sets up a domino effect. The anti-PE antibodies gain the opportunity to attack the cell; interfering with the cells’ signaling pathways and directing the cell into an inflammatory state, which ushers in greater risk of blood clot formation and pregnancy complications.
Funding for this work was provided by National Institutes of Health grants 5R01HL102085 and GM070736.
Early endosome as a pathogenic target for antiphosphatidylethanolamine antibodies. Songwang Hou, Heike Fölsch, Ke Ke, Joan Cook Mills, Rosalind Ramsey-Goldman, and Ming Zhao. Proceedings of the National Academy of Sciences USA. 2017 Dec 26;114(52):13798-13803. doi: 10.1073/pnas.1714027115. Epub 2017
Written by Ming Zhao, PhD