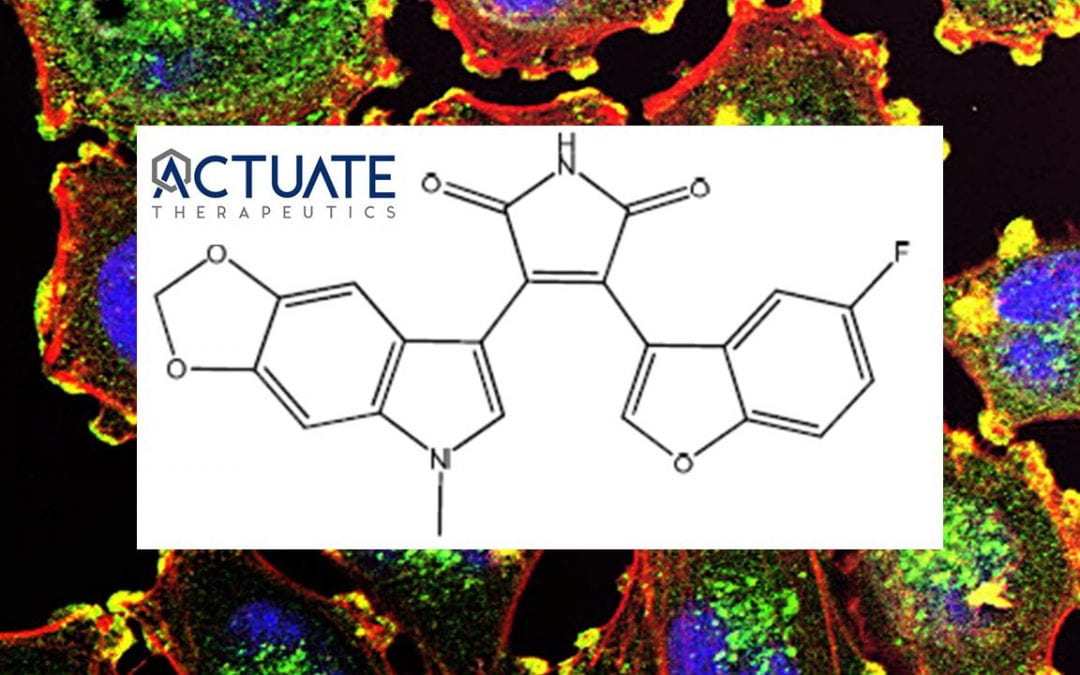
Veteran entrepreneurs and cofounders of Actuate Therapeutics, Inc., Daniel Schmitt and Andrew Mazar, PhD, are taking no chances with their lead clinical candidate 9-ING-41, a promising new treatment for advanced and drug-resistant cancers and select inflammatory diseases.
“Having done this many times before, we knew we needed a certain body of data to interest a potential pharma partner or investor,” said Mazar, the startup’s scientific co-founder. “All of our work was geared toward generating a proof of concept package.”
Mazar and Schmitt, President and CEO of Actuate, met at Northwestern as Entrepreneurs-In-Residence for Chemistry of Life Processes Institute’s (CLP) Center for Developmental Therapeutics (CDT) and the University’s Innovation and New Ventures Office (INVO), respectively. Schmitt was on the lookout for promising technologies to spin out. As EIR and managing director of CDT and faculty director of the center’s Developmental Therapeutics Core (DTC), Mazar led early academic drug discovery projects and developed commercialization strategies for the most promising programs. The core specializes in proof-of-concept study design and development of project packages for review by a pharmaceutical partner or investor. Through the generosity of the Baskes family philanthropy, CDT developed a repository of Patient Derived Xenografts (PDX) models used to assess new oncology drugs.
In 2015, DTC signed a cooperative research agreement with the University of Illinois-Chicago to run preclinical studies to test the efficacy of glycogen synthase kinase-3β (GSK-3β) inhibitors developed by UIC medicinal chemist Alan Kozikowski. GSK-3β is the gatekeeper of cell signaling pathways. Overexpression of this protein is found in Alzheimer’s disease, bipolar disorder, diabetes mellitus type II, and inflammatory conditions leading to fibrosis. When dysregulated, it allows tumor cells to proliferate without control and is a marker of malignancy. These characteristics make it an excellent target for drug therapies.
“The early work on this was really pointed towards neurodegenerative disease,” said Schmitt. Of all the compounds initially tested, the researchers found 9-ING-41 the most interesting in terms of specificity, potency, and brain penetration.
“One of the first models we looked at was primary malignant brain cancer (glioblastomas) and we saw this remarkable activity,” said Mazar. “When we started seeing this ability to restore sensitivity to chemotherapy that had been lost before, we got really excited.” Collaborations between Chemistry of Life Processes Institute, CDT, and DTC faculty and staff, and other Northwestern investigators resulted in approximately fifteen publications.
The discovery prompted Schmitt and Mazar to spin out the technology and know-how from UIC and Northwestern in 2015 and form Actuate. They also secured initial funding to complete preclinical studies.
An early pioneer in using PDX models of cancers to screen drugs, CDT began in vivo and in vitro studies of 9-ING-41 to test its efficacy in combination with different chemotherapy drugs in drug-sensitive and drug-resistant carcinomas. “The studies revealed significant increases in overall survival. The compounds were also well tolerated and demonstrated a favorable pharmacokinetic profile,” said Schmitt.
In March, 2016, the Federal Drug Administration granted 9-ING-41 an Orphan Drug Designation for treatment of glioblastoma. The following year, the company closed on $3.8 million in Series A funding for toxicology studies and the FDA further granted 9-ING-41 Orphan Drug Designation for the treatment of neuroblastoma in adults and children. In February 2018, the FDA accepted Actuate’s IND application for 9-ING-41, clearing the way for studies in patients with advanced cancers.
A Phase I/II clinical trial opened up in December, 2018, for treatment of chemo-resistant patients with advanced cancers, including glioblastoma, melanoma, pancreatic, appendix, breast and ovarian. The trial was bolstered by an additional $21.7 million in Series B funding allowing Actuate to expand the clinical program. Since initiation, more than 70 patients have been enrolled in the study. A recent influx of $6.5 million will enable Actuate to open up two additional clinical trials, one in myelofibrosis and one in pediatric neuroblastoma, with collaborators at 20 different clinical sites in the US and Europe.
“We’ve dose-escalated 9-ING-41 in clinical trials through six different dose levels and we’ve had no serious adverse events. We’re getting very good data on clinical responses to the drug so far,” said Schmitt. “Metastatic disease has stabilized in a number of patients, including one that has, to date, shown a durable and complete response without recurrence.”
Alicia Löffler, PhD, the Executive Director of INVO, who recruited Dan Schmitt as EIR and helped broker the startup’s licensing deal and subsequent financing, also is very encouraged. “Actuate’s progress is incredibly exciting,” says Löffler. ”Financial partners Kairos Ventures and others understand the importance and urgency in supporting early-stage innovations like 9-ING-41 that give new hope to millions of people living with the most aggressive forms of cancer.”
If things continue to go positively, Actuate aims to raise additional money to take the drug forward into registration, or to be acquired.
“I’ve been doing this for 30 years and what gets me up in the morning is knowing that we’re doing the right things to advance treatment for the patients,” said Schmitt. “That’s what we’re working for.”
by Lisa La Vallee